Measuring pH
Course Details
Premier
pH is one of the most commonly determined parameters of aqueous (and non-aqueous) solutions because many chemical processes are dependent on pH. Altering pH can often significantly alter the rate of chemical reactions. The solubility and bioavailability of many pharmaceutically active compounds is also determined by solution pH. Furthermore, in chromatography, the retention and selectivity of ionizable analytes can be manipulated by changing the eluent pH. This module will discuss the equipment used to measure pH and how to use it.
Topics include:
- Introduction and theory
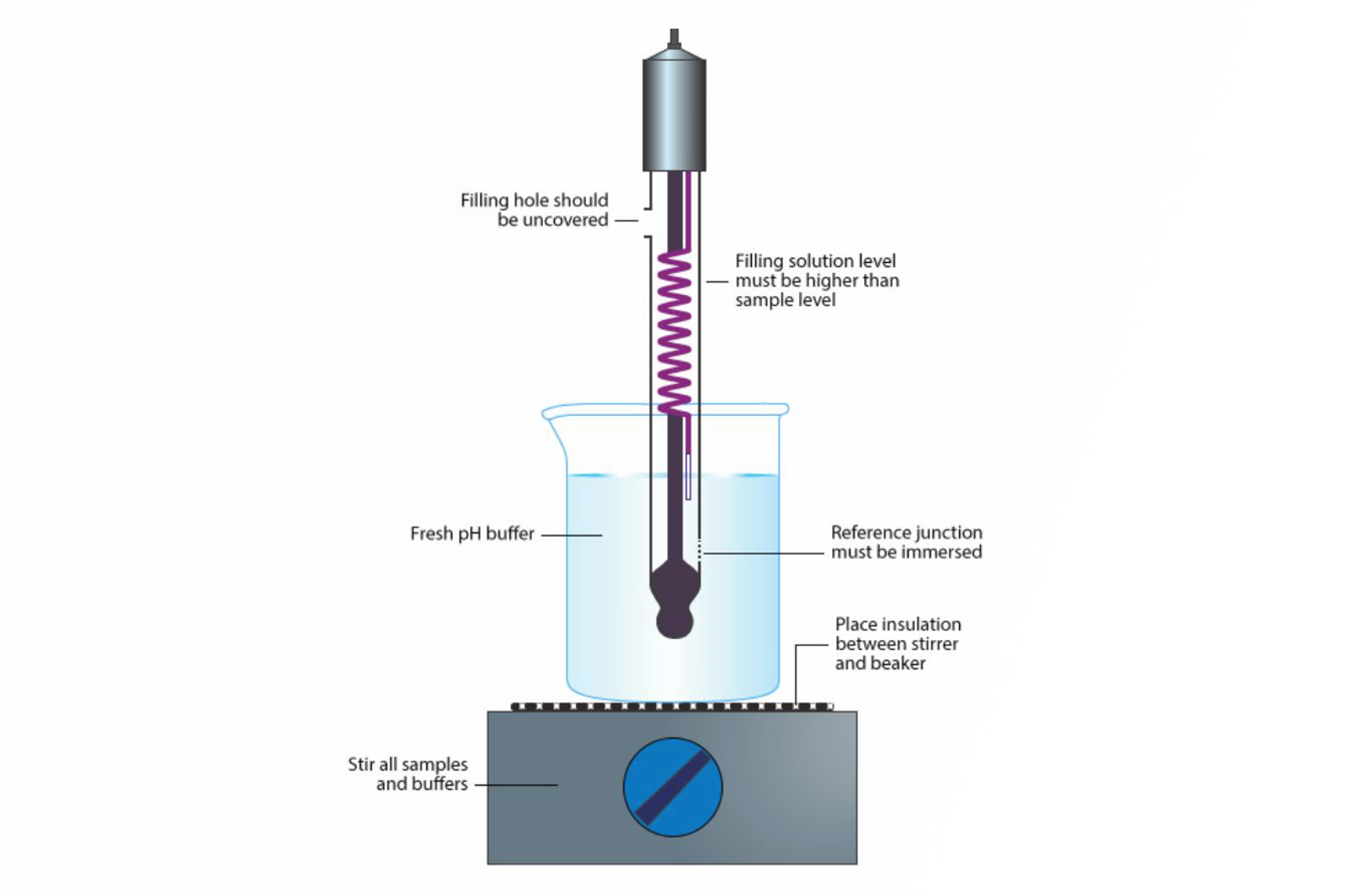
- pH paper and pH meters
- pH meter electrodes
- Buffer solution and pH electrode characteristics
- Correct use of electrodes, electrode care, and maintenance
- Calibrating a pH meter and making a measurement
- Buffer preparation using the Henderson-Hasselbalch equation
- Buffer preparation from stock solutions
- Calculating the masses of buffer components
- Calculating the pH of a strong acid solution
- Calculating the pH of a strong base solution
- Calculating the pH of a weak acid solution
- Calculating the pH of a weak base solution
- pH of common acids and bases