Titrations
Course Details
Premier
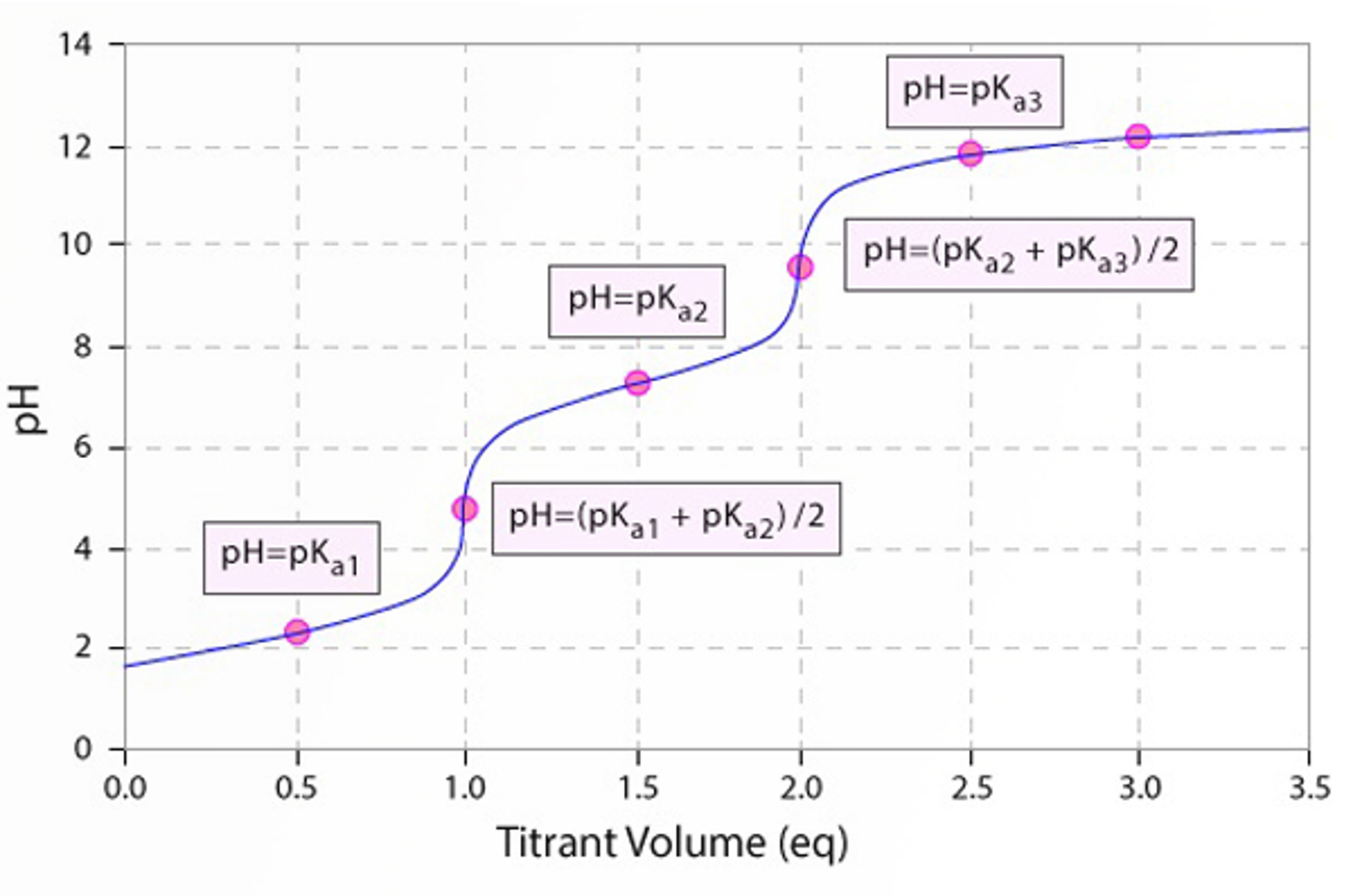
A titration is the determination of the specific quantity of an analyte by the addition of a reagent (titrant) of known concentration. The reaction involved in a titration must be fast, complete, unambiguous, and observable. The titrant is added until the reaction is complete, at which point the so-called end point, or equivalence point, is reached. The end point of the reaction is monitored using a color change indicator, pH meter, or electrochemical sensor. The volume of titrant added allows the quantity or concentration of the analyte to be calculated based upon the reaction stoichiometry. This module will discuss the different types of titrations, how to perform a titration, and how to overcome common problems.
Topics include:
- Titrations
- Acid-base titrations
- Titrant standardization
- Indicators for acid-base titrations
- Titration of a weak acid with a strong base
- Titration of a weak base with a strong acid
- How to carry out a titration
- Glossary of titration terms
- Errors in titration
- Moisture determination (Karl Fischer titration)
- Karl Fischer titration, side reactions, and rate control
- Bivoltametrc indication
- End point polarization
- Drift
- Calibration,. maintenance, and troubleshooting of Karl Fischer autotitrators
- Errors with Karl Fischer titrations