HPLC Techniques in Biopharmaceutical Analysis
Course Details
Biopharmaceuticals offer great hope in treating medical conditions which are currently poorly served at best by traditional pharmaceuticals. It is estimated that there are over 400 biopharmaceuticals in clinical trails for in excess of 200 disease areas. The enhanced complexity and variability that comes from the size of biopharmaceuticals, allied with the intricacy of the production process, mean chromatography is employed to a much greater extent during production and release testing. This webcast will provide details of why differing modes of chromatography are required in the research, preparation and analysis of biopharmaceuticals and when they would be typically used.
Topics include:
- Introduction to biopharmaceuticals
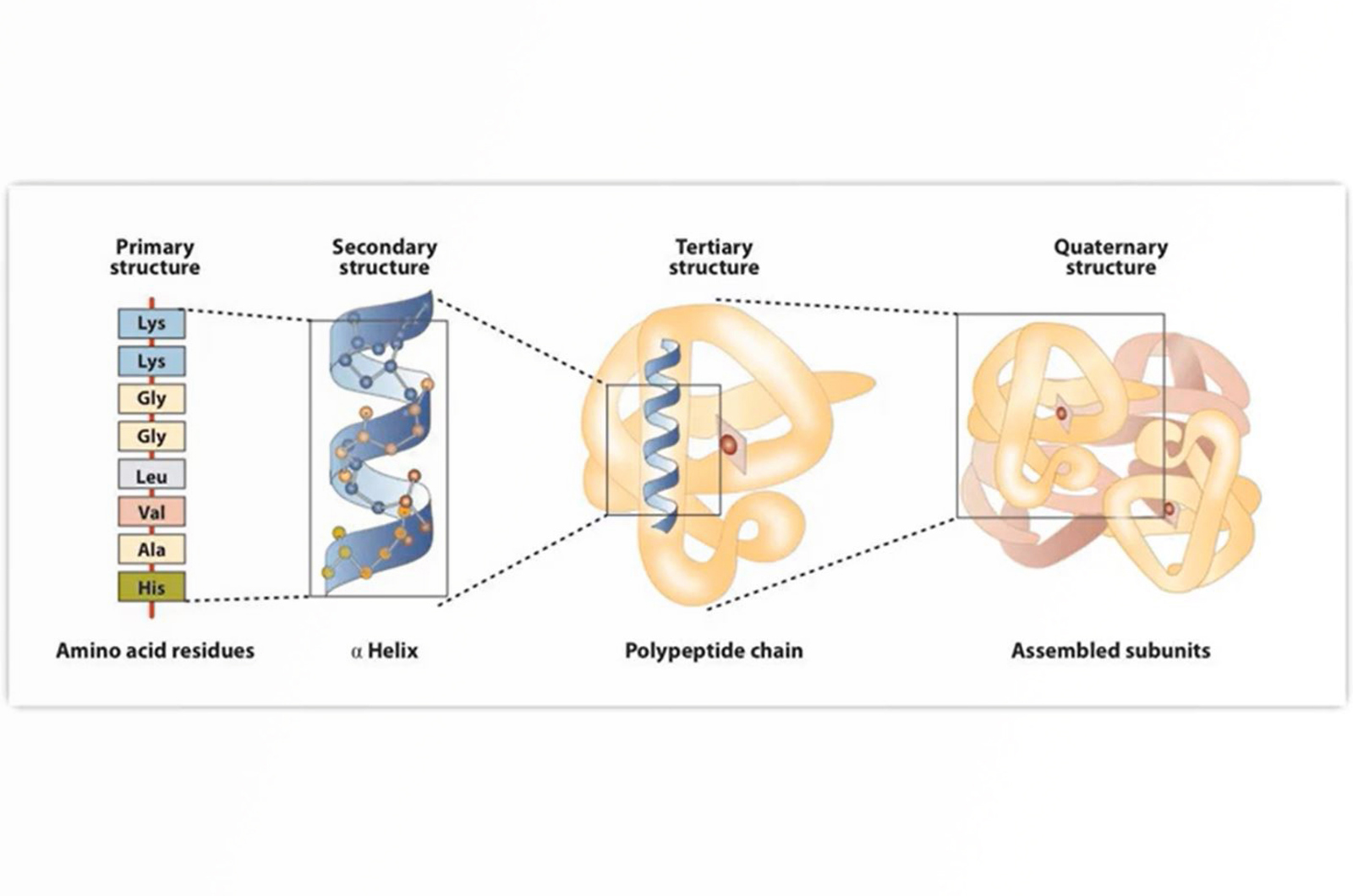
- Protein characteristics of analytical interest
- Use of various HPLC techniques to analyze proteins at intact (native), fragmented, peptide, glycan and amino acid levels including:
- Reversed phase (RPLC)
- Size exclusion chromatography (SEC)
- Ion exchange chromatography (IEX)
- Hydrophilic interaction chromatography (HILIC)
- Affinity chromatography
- Advances in throughput, specificity and accuracy for peptide mapping, charged variant analysis, aggregation, and titer analysis