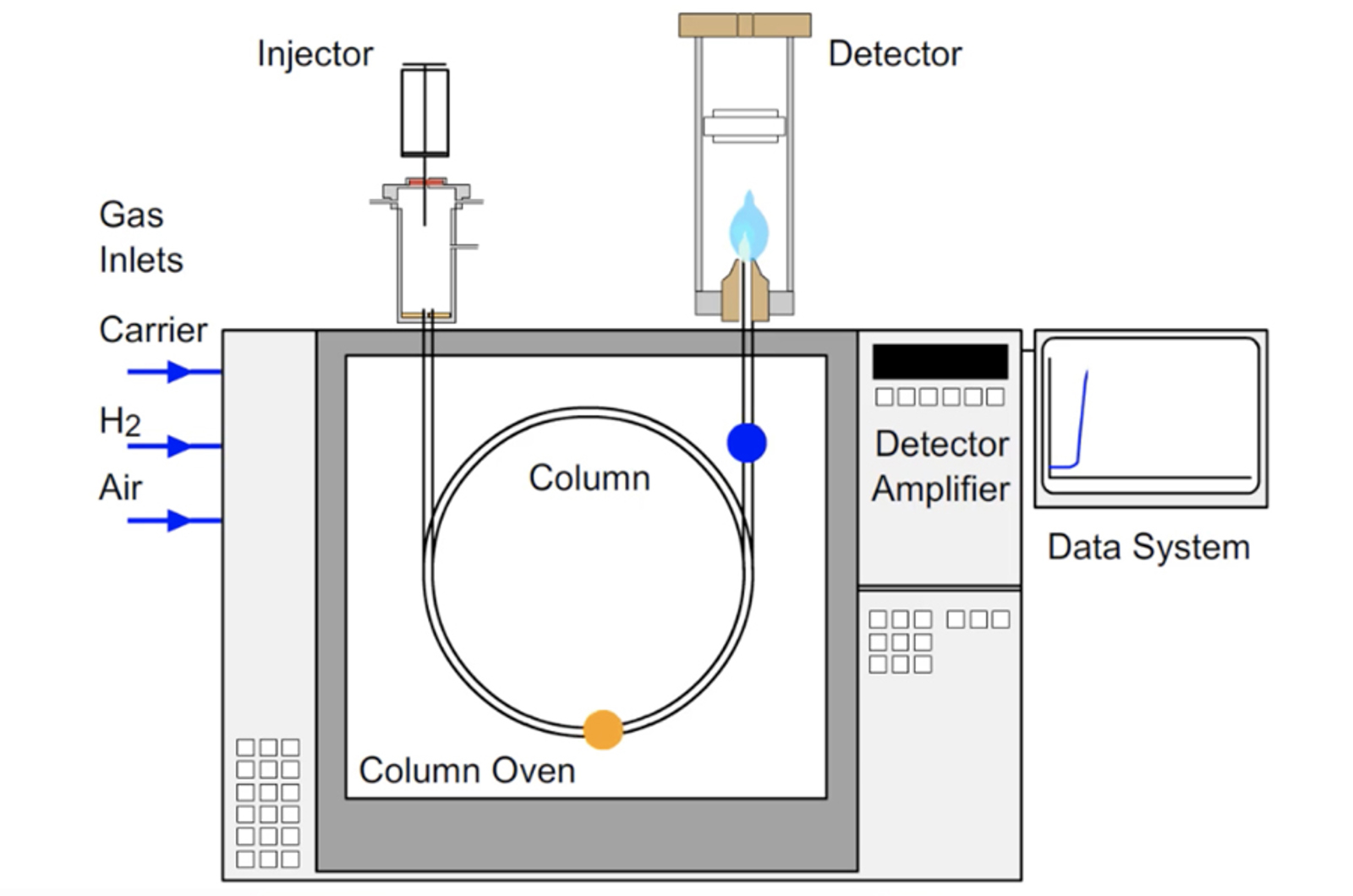
GC Method Development Video Training Course
Endorsed by

This course is approved by the Royal Society of Chemistry & American Chemical Society - Analytical Division.

Method development in the modern GC laboratory is challenging but can be approached very logically and is hugely rewarding when a fit for purpose method is produced. This course provides you with the underpinning knowledge to begin your GC method development career.
This course has been designed for those who are experience in GC and who are now looking to develop their experience and understanding of method development. During the course we will assume that you are familiar with the basic theories of gas phase chromatographic separations, but we’ll also revisit some theory to underpin the teaching.
Logical and informed method development leads to high quality, reproducible and robust GC methods. This course considers the wide array of equipment and chemistry choices available to method developers in the modern analytical laboratory and teaches a refined and logical approach to method development.
Each video session is accompanied by supporting modules from CHROMacademy and we will be using some of the thousands of movies within CHROMacademy to clearly demonstrate the principles and processes being discussed.
All four sessions should be attended in order to gain the level of understanding required in order to complete the Final Assessment, which is a mandatory element required in order to gain your certificate of course completion.
Session 1:
We begin by discussing how to set analytical objectives, because, in the words of the Wizard of Oz, if you don’t know where you are going, how do you know when you’ve arrived! This will involve assessment of the sample matrix and analyte chemistry (where known) and their influence on designing methods and setting analytical objectives.
We’ll look at the sample preparation options available to us and the types of preparation that might be required for various inlet types and detection techniques. We also begin our discussion on selecting the correct mode of chromatography for our separation and examine some of the stationary phase chemistries available to today’s method developers.
Session 2:
In our second session we begin to explore the choices available for sampling and sample introduction. We discuss in-depth the choices for split and splitless injection and the important variables associated with each of these techniques.
We will consider important choices such as inlet liner design and selection, factors which determine split ratio, and unique splitless injection parameters such as initial oven temperature and purge time.
We will also consider how to avoid insidious issues such as discrimination and backflash. In this session we will also investigate headspace analysis and other sampling techniques.
Session 3:
In this session we investigate the important choices which govern the selection of GC column dimensions and how to optimize efficiency and analysis time.
We also discuss sample screening methods to investigate the retention behavior of the analytes and make some initial decisions regarding the requirements for the temperature program.
Using temperature programming theory, we investigate how to optimize the temperature program parameters, such as initial and final temperature and the slope of the temperature gradient. We discuss the use of mid ramp holds for separating critical peak pairs within the analysis. We also begin our discussion on GC detectors by examining the range of detector types available and the factors which govern detector selection.
Session 4:
In the final session we take an in-depth look at various popular detectors for GC and highlight the important variables which govern response and dictate sensitivity.
We look at the full range of ionizing detectors, including mass spectrometric detectors as well as electron capture and thermal conductivity detectors and highlight the key method development decisions associated with each detector.
We conclude the course with an example method development which will serve to reinforce your learning and highlight the key method development decisions and optimization processes associated with logical method development for capillary GC.