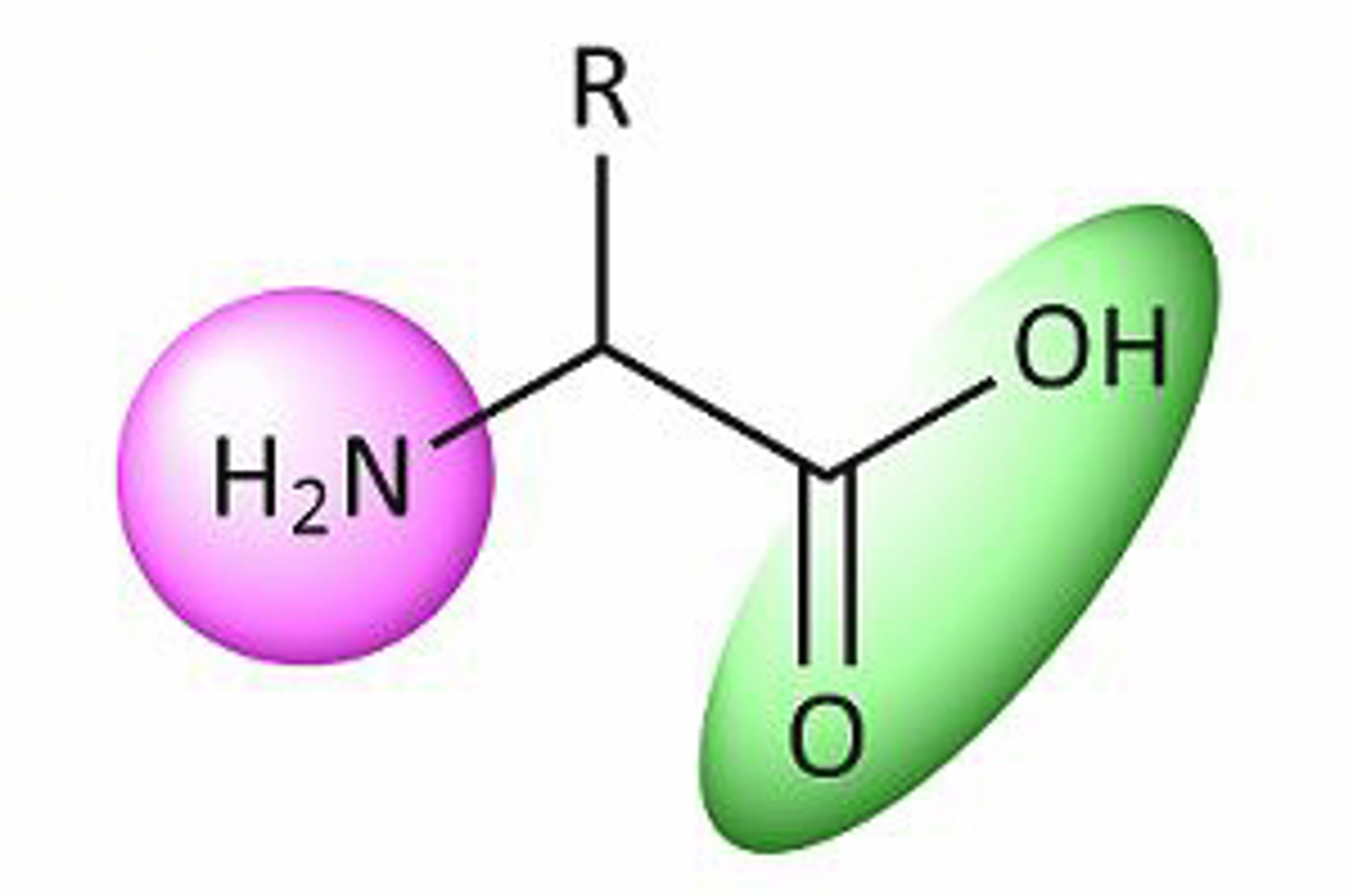
Reversed Phase HPLC for the Analysis of Biomolecules
Course Details
Quick Guide
Premier
Author: Crawford Scientific
The following article will introduce the fundamentals of biopharmaceutical analysis and cover the use of reversed phase HPLC in their analysis