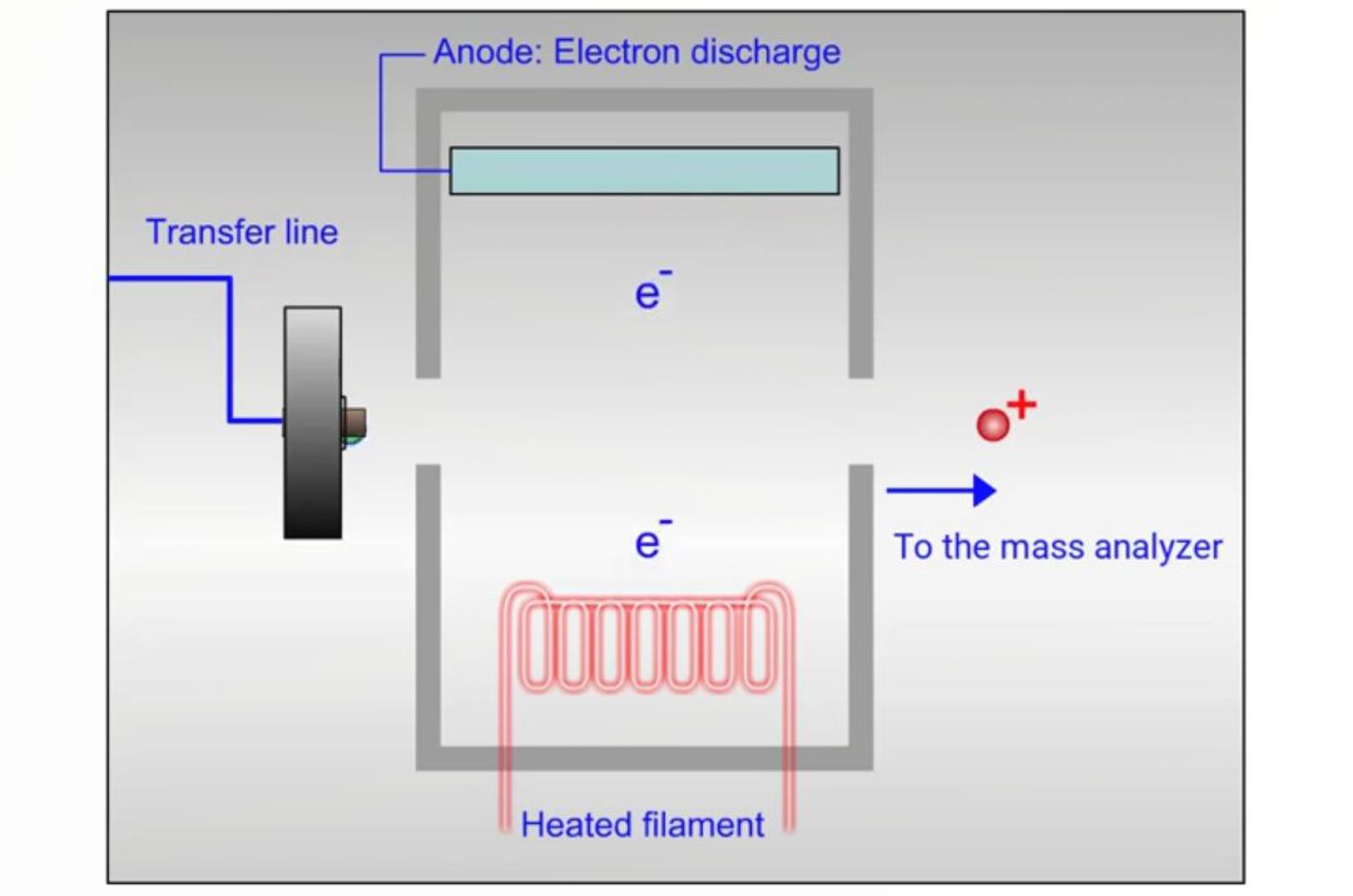
Fundamentals of GC-MS Ionization Techniques
Course Details
Premier
This quick guide gives a concise overview of GC-MS ionization technique.