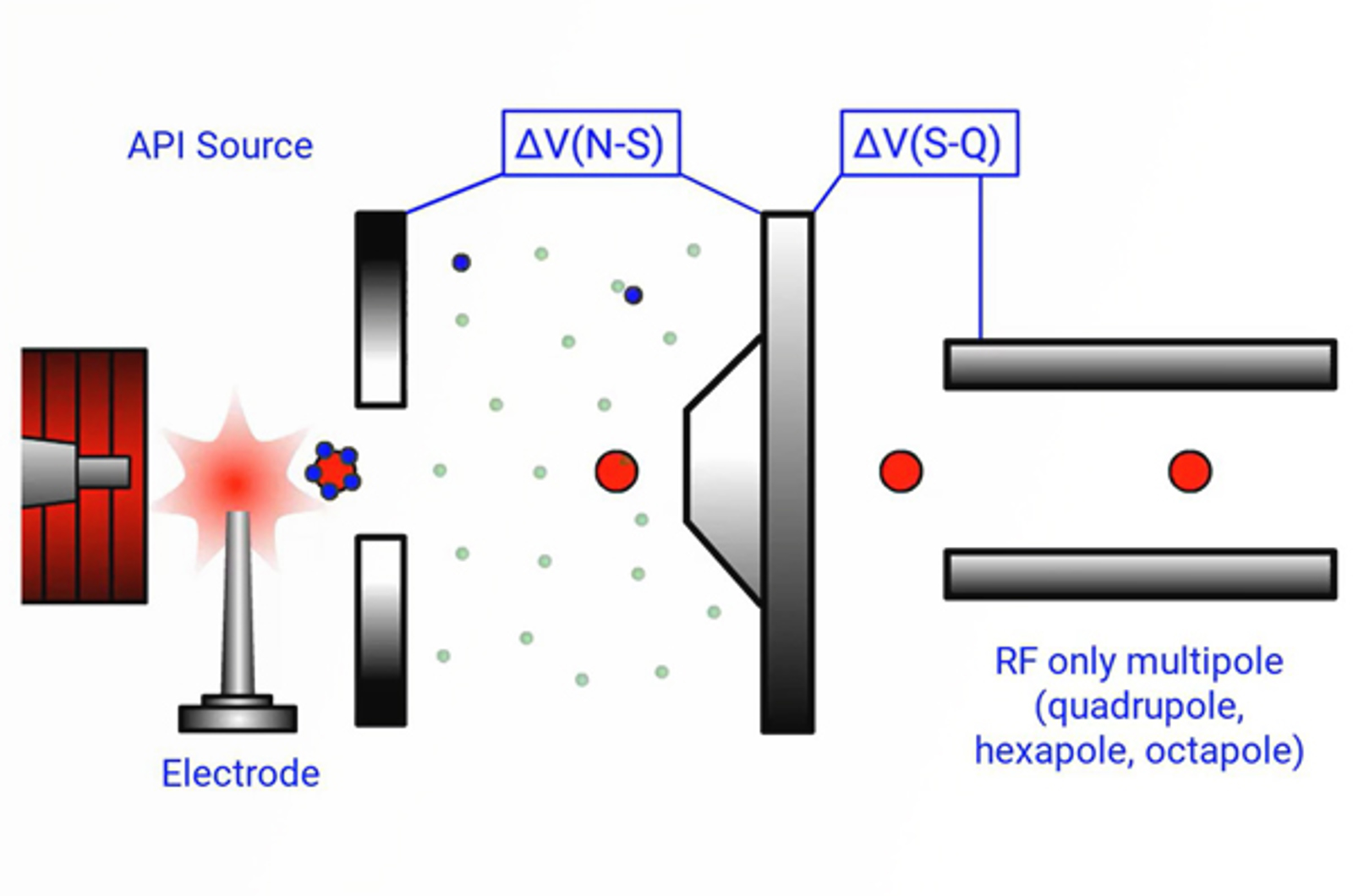
7 Must Know Points to Understand Atmospheric Pressure Chemical Ionization
Course Details
Quick Guide
Premier
Here is our 7 point guide to understanding the mechanism of atmospheric pressure chemical ionization (APCI).