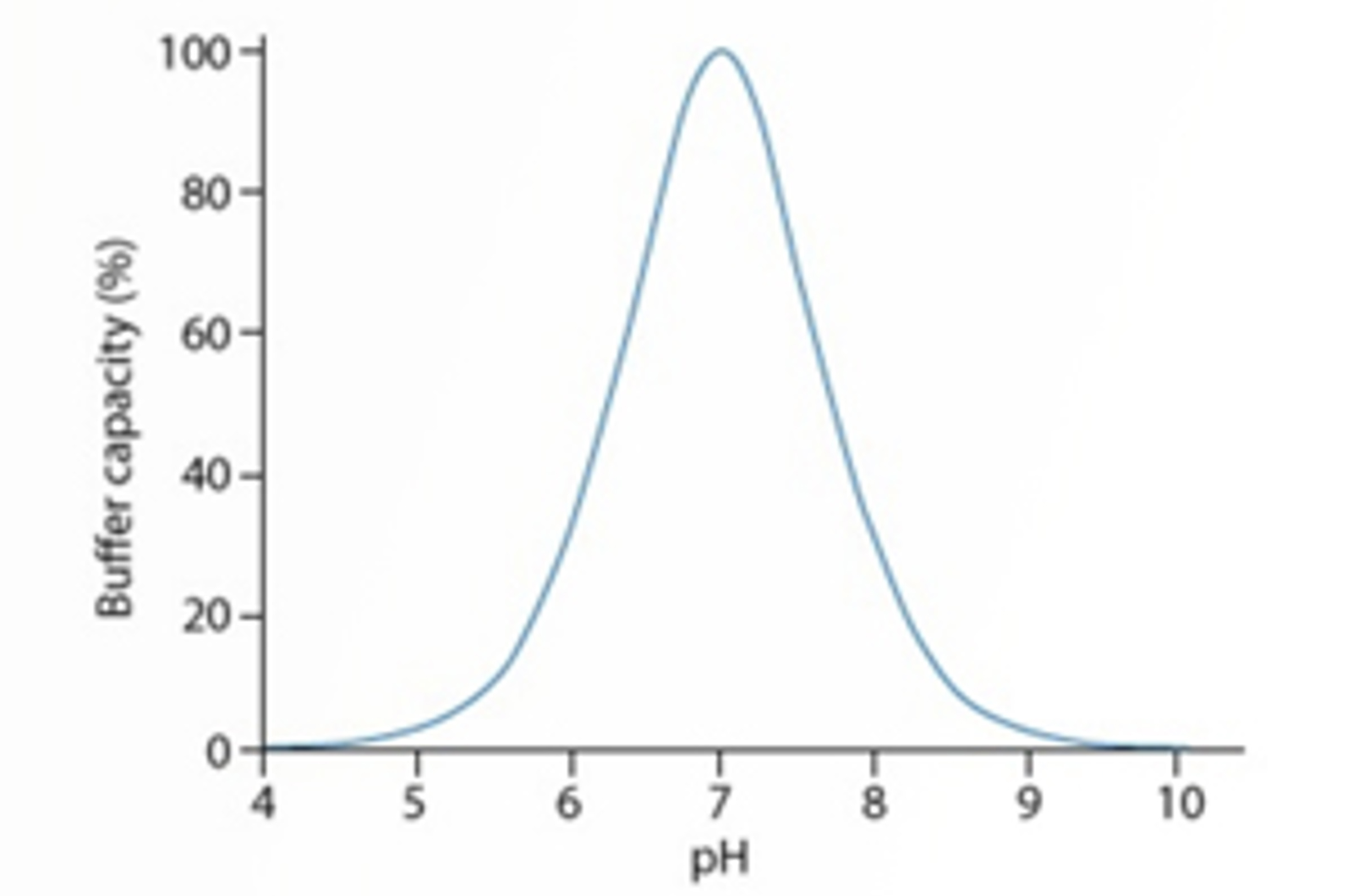
Buffer Choice for HPLC Separations
Course Details
Quick Guide
Premier
This quick guide looks at the important topic of buffer choice for HPLC separations; how to the choose the correct buffer type and concentration, as well as how to avoid variability in retention and selectivity.